Update to ASCO/CAP Guideline in Breast Cancer: No Sweeping Changes, but Many Refinements - Cancer Therapy Advisor

2018 ASCO/CAP summary recommendations [original recommendations and... | Download Scientific Diagram
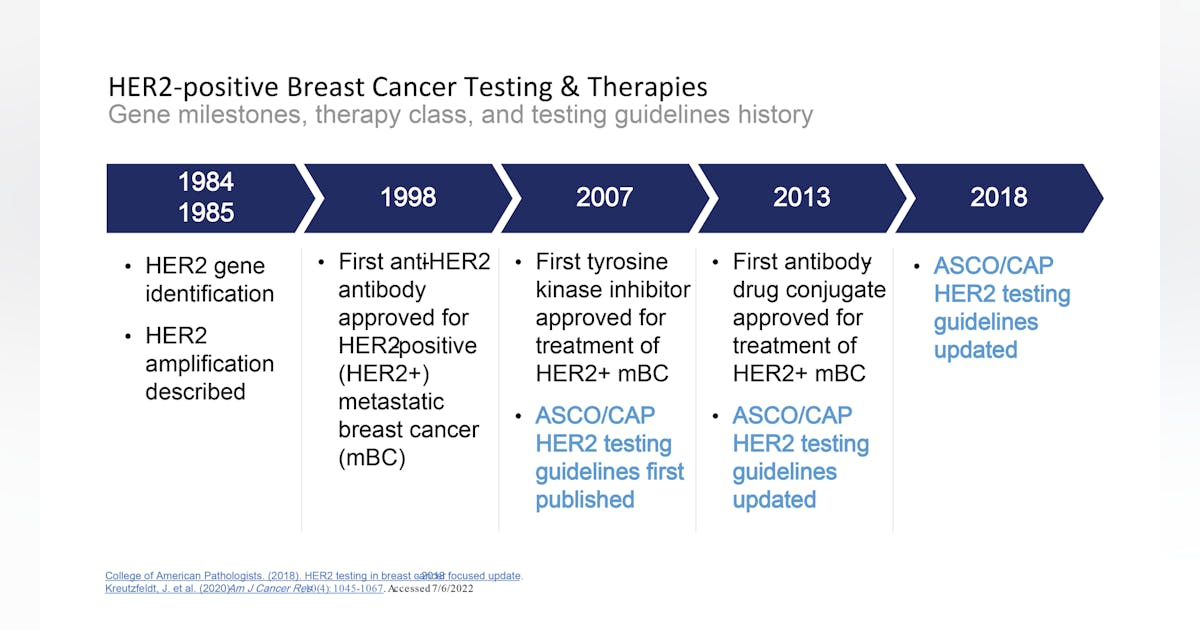
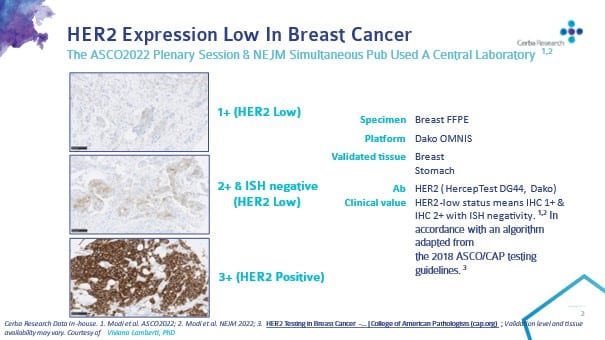
Breast cancer biomarkers, and a new clinical category for HER2 expression | Medical Laboratory Observer

HER2 spectrum of expression according to ASCO/CAP guidelines. HER2-low... | Download Scientific Diagram

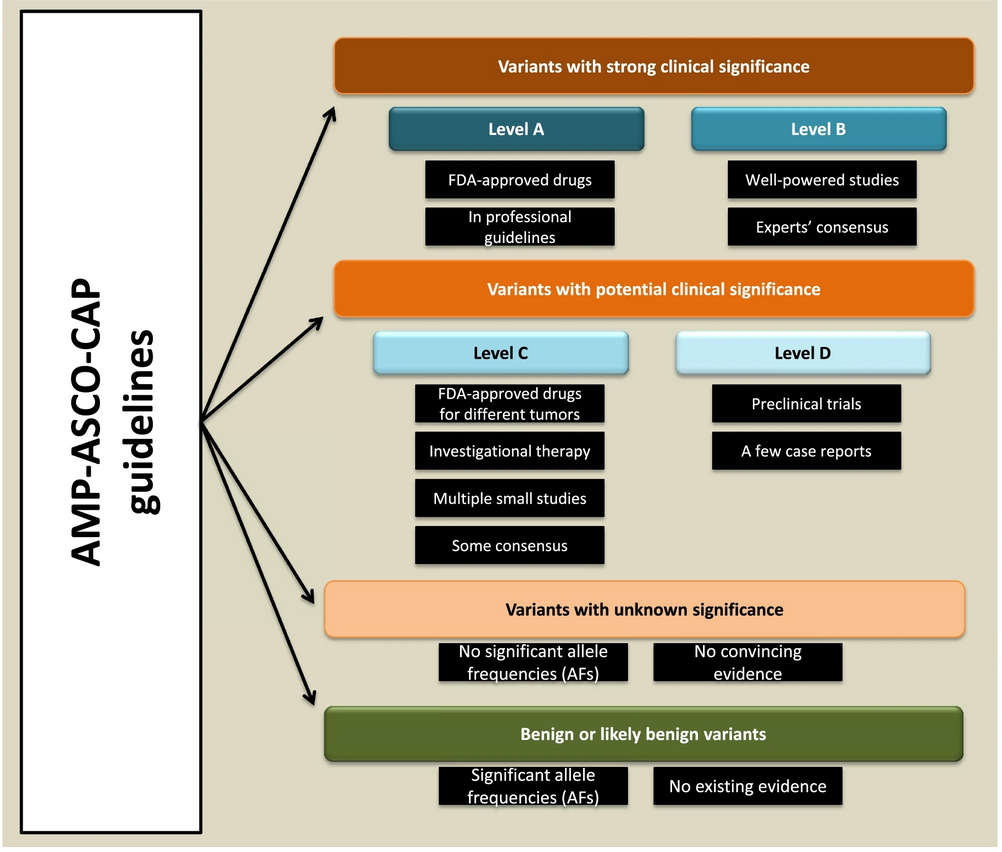
Clinically actionable cancer somatic variants (CACSV): a tumor interpreted dataset for analytical workflows | BMC Medical Genomics | Full Text

Clinical significance of quantitative categorization of HER2 fluorescent in situ hybridization results in invasive breast cancer patients treated with HER2-targeted agents - Modern Pathology

PPT - HER2 Testing in Breast Cancer: 2013 ASCO/CAP HER2 Guideline Update PowerPoint Presentation - ID:6109259

Estrogen and Progesterone Receptor Testing in Breast Cancer: ASCO/CAP Guideline Update | Journal of Clinical Oncology
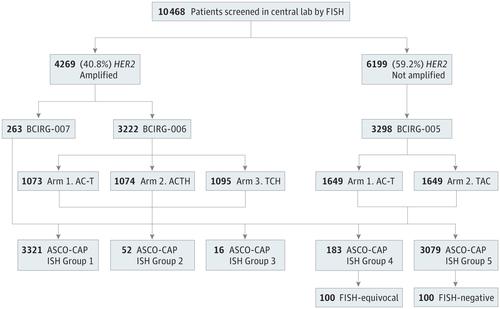
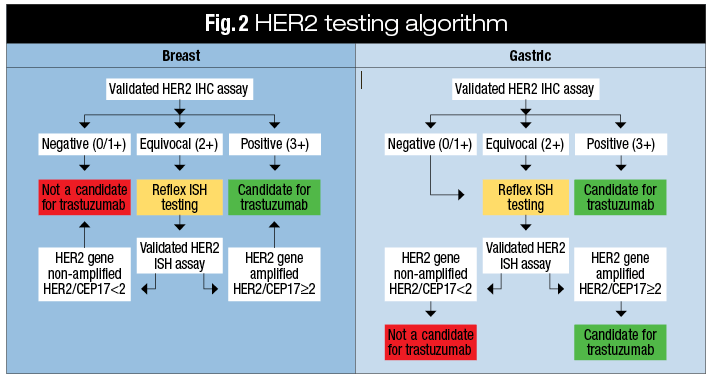
Assessment of ERBB2/HER2 Status in HER2-Equivocal Breast Cancers by FISH and 2013/2014 ASCO-CAP Guidelines.,JAMA Oncology - X-MOL
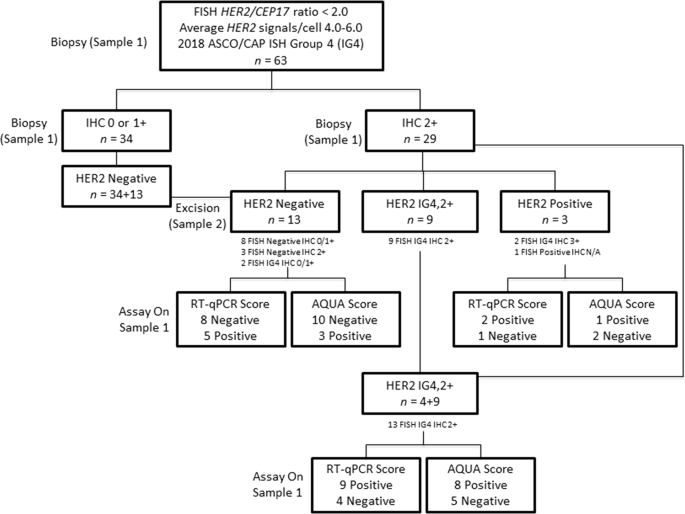
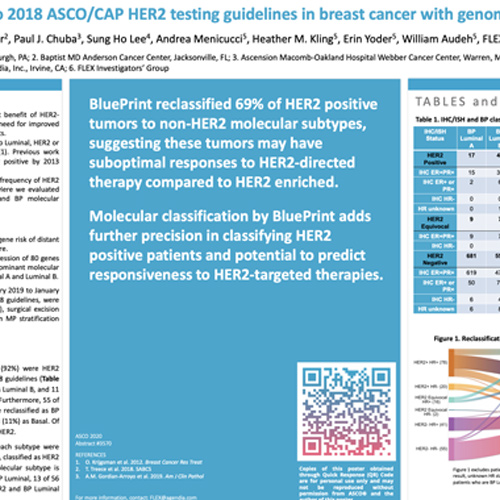
Quantitative assessments and clinical outcomes in HER2 equivocal 2018 ASCO/ CAP ISH group 4 breast cancer | npj Breast Cancer
![PDF] HER2 status in breast cancer: changes in guidelines and complicating factors for interpretation | Semantic Scholar PDF] HER2 status in breast cancer: changes in guidelines and complicating factors for interpretation | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/b66331181b2eafa14b6e478dcc7fced0c7917993/3-Table1-1.png)
PDF] HER2 status in breast cancer: changes in guidelines and complicating factors for interpretation | Semantic Scholar
![PDF] THE BOTTOM LINE Human Epidermal Growth Factor Receptor 2 Testing in Breast Cancer : American Society of Clinical Oncology / College of American Pathologists Clinical Practice Guideline Focused Update Guideline Questions | Semantic Scholar PDF] THE BOTTOM LINE Human Epidermal Growth Factor Receptor 2 Testing in Breast Cancer : American Society of Clinical Oncology / College of American Pathologists Clinical Practice Guideline Focused Update Guideline Questions | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/747f72db6685fa520b07391fb016bac6754d7633/9-Figure3-1.png)
PDF] THE BOTTOM LINE Human Epidermal Growth Factor Receptor 2 Testing in Breast Cancer : American Society of Clinical Oncology / College of American Pathologists Clinical Practice Guideline Focused Update Guideline Questions | Semantic Scholar